Math & Physics
Respiratory¶
Gas Laws¶
Ideal Gas Law¶
Pressure, Volume, number of moles, universal gas constant, temperature
Boyle's Law¶
This law describes an inverse relationship. An example is the diaphragm contracting or shortening and increasing tidal volume.
Charles' Law¶
Gay Lussac's Law¶
Henry's Law¶
At a constant temperature the amount of gas that dissolves in the solution is directly proportional to the partial pressure of the gas over the solution...aka higher the gas pressure the more dissolves (overpressurizing the vaporizer). But if temp increases, then the solubility goes down and vice versa...so dec temp inc solubility and therefore prolongs emergence since the gasses are not just staying in the lungs or in circulation. Henry’s constant is more of a coefficient since its value is temperature dependent.
Dalton's Law¶
Alveolar Gas Equation¶
$ P_a $ is Atmospheric Pressure and $ P_w $ Pressure of water vapor
Dead Space¶
Bohr Equation
Spontaneous Ventilation
Controlled Ventilation
Increased \(V_D\) due to decreased BP
Respiratory Quotient Derivation¶
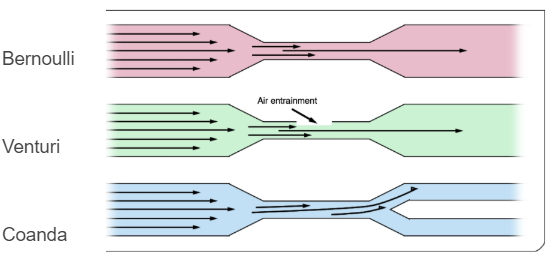
Fluid Dynamics Review¶
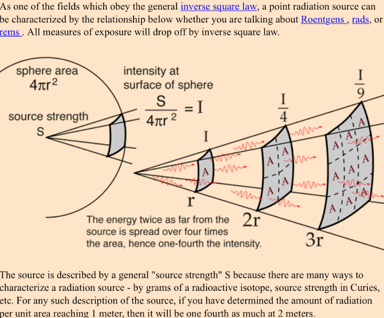
Inverse Sqare Law and Radiation Safety¶
Time Constant and the Circuit¶
Volume of the circuit is a known constant and the total fresh gas flow FGF is Q. Three time time constants τ are required for 95% change in concentration. So as the FGF is decreased the time to equillibration is increased.